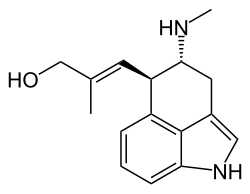
Chanoclavine
Appearance
 | |
| Names | |
|---|---|
| IUPAC name
[9(9a)E]-9-Methyl-9,9a-didehydro-7,8-seco-9a-homoergolin-8-ol
| |
| Systematic IUPAC name
(2E)-2-Methyl-3-[(4R,5R)-4-(methylamino)-1,3,4,5-tetrahydrobenzo[cd]indol-5-yl]prop-2-en-1-ol | |
| Other names
chanoclavin-l
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H20N2O | |
| Molar mass | 256.34 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chanoclavine, also known as chanoclavin-I, is a tricyclic ergot alkaloid (ergoline) isolate of certain fungi. It is mainly produced by members of the genus Claviceps.[1] Long used in traditional Chinese medicine, it was found in 1987 mouse studies to stimulate dopamine D2 receptors in the brain.[2] It is described as being devoid of ergot-like activity, possessing no outstanding pharmacological activity, and as not contributing to the hallucinogenic effects of morning glory seeds.[3][4][5][6][7][8]
See also
[edit]References
[edit]- ^ Lorenz, N; Haarmann, T; Pazoutová, S; Jung, M; Tudzynski, P (2009). "The ergot alkaloid gene cluster: Functional analyses and evolutionary aspects". Phytochemistry. 70 (15–16): 1822–32. doi:10.1016/j.phytochem.2009.05.023. PMID 19695648.
- ^ Watanabe, H; Somei, M; Sekihara, S; Nakagawa, K; Yamada, F (1987). "Dopamine receptor stimulating effects of chanoclavine analogues, tricyclic ergot alkaloids, in the brain". Japanese Journal of Pharmacology. 45 (4): 501–6. doi:10.1254/jjp.45.501. PMID 3127619.
- ^ Hofmann A (1963). "The Active Principles of the Seeds of Rivea Corymbosa and Ipomoea Violacea". Botanical Museum Leaflets, Harvard University. 20 (6). Harvard University Herbaria: 194–212. ISSN 0006-8098. JSTOR 41762231. Archived from the original on 28 March 2025.
[...] chanoclavine, which has no outstanding pharmacological activity, appears to play no part in the occurrence of the psychic effects of badoh and badoh negro.
- ^ Albert Hofmann (1968). "Psychotomimetic Agents". In Burger A (ed.). Drugs Affecting the Central Nervous System. Vol. 2. New York: M. Dekker. pp. 169–235. OCLC 245452885. OL 13539506M.
Furthermore, chanoclavine, which has no outstanding pharmacological activity, appears to play no part in the occurrence of the psychic effects of ololiuqui.
- ^ Brimblecombe RW, Pinder RM (1975). "Indolealkylamines and Related Compounds". Hallucinogenic Agents. Bristol: Wright-Scientechnica. pp. 98–144. ISBN 978-0-85608-011-1. OCLC 2176880. OL 4850660M.
d-Lysergic acid amide (ergine) is the major constituent of the seeds of both Rivea corymbosa and Ipomoea violacea, together with smaller amounts of d-isolysergic acid amide (isoergine), chanoclavine, elymoclavine, and the N-(1-hydroxyethyl)amides of lysergic and isolysergic acids. [...] It is clear that the pharmacologically active constituents of ololiuqui are the isomeric lysergic acid amides. [...] Heim and his colleagues suggest that the overall effects of ololiuqui are due to these two compounds, the d-lysergic acid amide giving intoxication with strong autonomic side-effects and the d-isolysergic acid amide producing some euphoria, synaesthesia, and altered time experience. Certainly elymoclavine, lysergol, chanoclavine, and ergometrine produce no psychic changes in man (Isbell and Gorodetzky, 1966; Hofmann, 1968), though the first two do produce central excitation in animals (Yui and Takeo, 1958).
- ^ Heacock RA (1975). "Psychotomimetics of the Convolvulaceae". Prog Med Chem. 11: 91–118. doi:10.1016/s0079-6468(08)70209-1. PMID 1078534.
No psychological effects have been reported for ergometrine (6), a drug widely used in obstetrics, nor for chanoclavine (3).
- ^ Fanchamps A (1978). "Some Compounds With Hallucinogenic Activity". In Berde B, Schild HO (eds.). Ergot Alkaloids and Related Compounds. Handbook of Experimental Pharmacology (HEP). Vol. 49. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 567–614. doi:10.1007/978-3-642-66775-6_8. ISBN 978-3-642-66777-0.
The last component, Chanoclavine, is a tricyclic alkaloid, which is devoid of ergot-like activities. [...] Table 2. Psychotomimetic activity and some pharmacodynamic effects of structural analogues of LSD [...]
- ^ Hofmann A (January–March 1971). "Teonanácatl and Ololiuqui, two ancient magic drugs of Mexico". Bulletin on Narcotics. 23 (1): 3–14. Archived from the original on 28 March 2025.
Furthermore, chanoclavine, which has no outstanding pharmacological activity, appears to play no part in the occurrence of the psychic effects of ololiuqui.
